Reactivity series wikipedia9/25/2023 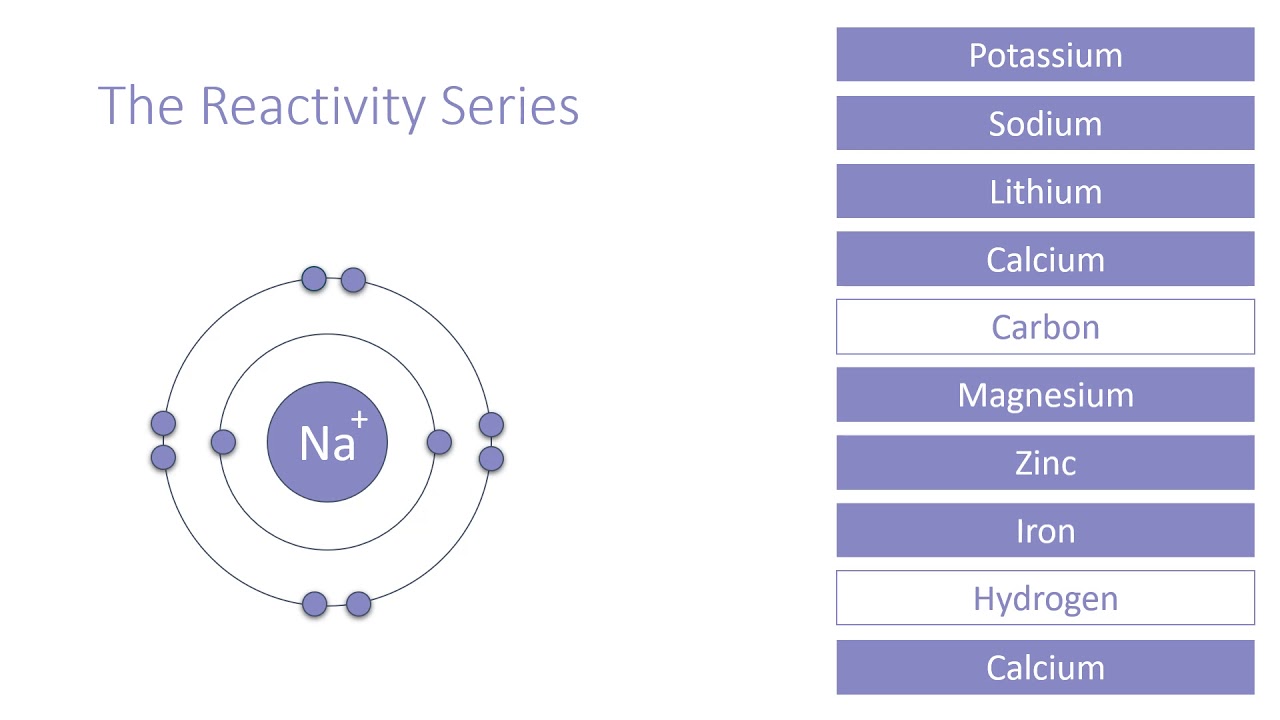
Classic pi-donor ligands are oxide (O 2−), nitride (N 3−), imide (RN 2−), alkoxide (RO −), amide (R 2N −), and fluoride. The existence of terminal oxo ligands for the early transition metals is one consequence of this kind of bonding. Their interaction is complementary to the behavior of pi-acceptor ligands. 
In coordination chemistry, a pi-donor ligand is a kind of ligand endowed with filled non-bonding orbitals that overlap with metal-based orbitals. Historically, CO and NO +Īre not included in this classification, nor are halides. N (n = 0, 1) where R is H or an organic substituent, or pseudohalide. The term 'metal ligand multiple bond" is often reserved for ligands of the type CR Symmetry arguments suggest that most ligands engage metals via multiple bonds. Furthermore, the usage of multiple bonding is not uniform. Most common classes of complexes showing metal ligand multiple bondsĪs a cautionary note, the classification of a metal ligand bond as being "multiple" bond order is ambiguous and even arbitrary because bond order is a formalism. Metal oxo intermediates are pervasive in oxidation catalysis. Transition metal carbene complexes catalyze the olefin metathesis reaction. Coordination complexes featuring multiply bonded ligands are of both scholarly and practical interest. In organometallic chemistry, a metal–ligand multiple bond describes the interaction of certain ligands with a metal with a bond order greater than one. 
Chemical interaction of certain ligands with metals of bond order >1
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
